Latest News
GLP-1 Drugs Enhance Success Rates for Kidney Transplants
GLP-1 receptor agonist drugs, originally developed for diabetes and weight loss, are now showing promise in improving outcomes for kidney…
Injectable Drug Xolair Effectively Reduces Severe Food Allergies in One-Third of Children
A groundbreaking study has revealed that the injectable drug Xolair (omalizumab) can significantly reduce the severity of food allergies…
New Medicare Rules Aim to Cut Radiation Exposure From CT Scans
The Centers for Medicare & Medicaid Services (CMS) have introduced new regulations aimed at reducing radiation exposure from CT scans..

Drug Application (NDA) is the vehicle through which drug sponsors
The New Drug Application (NDA) is the vehicle through which drug sponsors formally propose that the FDA approve a new pharmaceutical for sale and marketing in the United States.The purpose of a NDA is to provide enough information to permit the FDA to reach the following key decisions Whether the drug is safe and effective in its proposed use(s), and whether the benefits of the drug outweigh the risks Whether the drug’s proposed labeling (package insert) is appropriate and what it should contain Whether the methods used in manufacturing the drug and the controls used to maintain the drug’s quality are adequate to preserve the drug’s identity, strength, quality, and purity For more information on new drug applications, please visit the FDA’s How drugs are developed and approved page. NRX-100 (ketamine) Company: NRx Pharmaceuticals, Inc.Treatment for: Major Depressive Disorder with Acute Suicidal Ideation NRX-100 (ketamine) is an intravenous formulation of ketamine in development for the treatment of suicidal depression. NRx Pharmaceuticals, Inc. Files Initial Section of U.S. New Drug Application to the FDA for NRX-100 (IV Ketamine) for the Treatment of Suicidal Depression – December 30, 2024 See also: Generic approvals, New drug approvals, Recent additions to Drugs.com, Alphabetical listing of all new drug applications, FDA approval process Avutometinib Company: Verastem

Drugs Recently Received FDA approval, Newly approved medications
The following drugs have recently received FDA approval, including newly approved medications and new indications for existing drugs.

Updated on new drug approvals and regulatory processes
resources for staying updated on new drug approvals and regulatory processes..
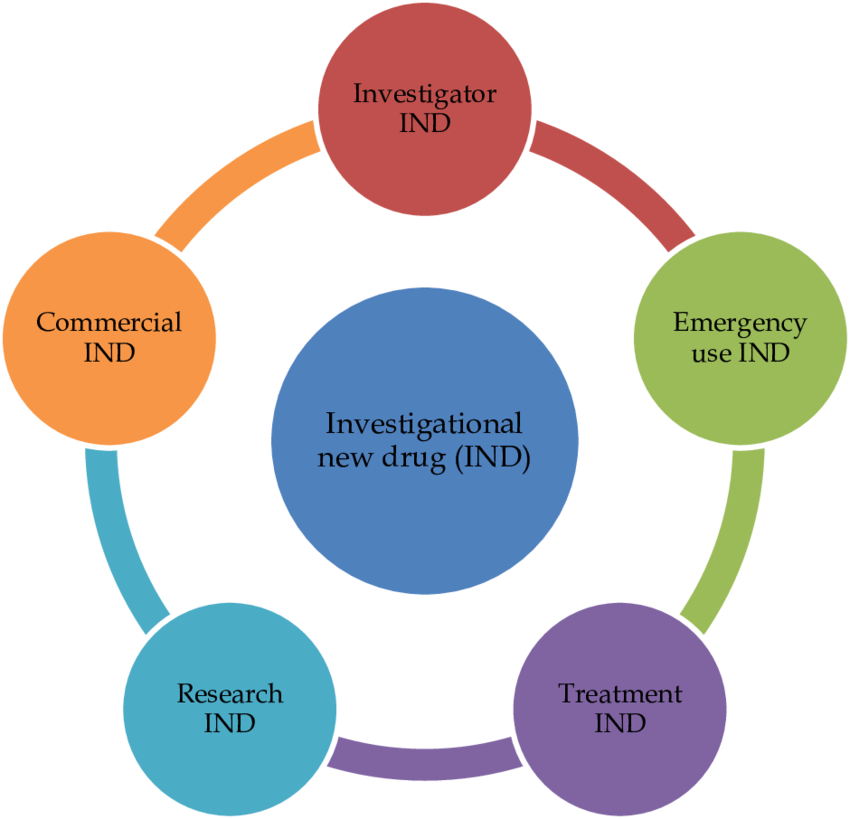
New drug approvals, regulatory agencies like the U.S. FDA categorize applications
When it comes to new drug approvals, regulatory agencies like the U.S. FDA categorize applications into different types based on the nature of the drug, its intended use, and prior approvals….
FDA approval, including newly approved medications and new indications
The following drugs have recently received FDA approval, including newly approved medications and new indications for existing drugs…
How Drugs Are Developed and Approved in the U.S.A
The process of drug development and approval in the U.S. is regulated by the Food and Drug Administration (FDA) and consists of several key stages to ensure the drug is safe and effective…
Azithromycin is a broad-spectrum antibiotic used to treat
Azithromycin 500mg is a broad-spectrum antibiotic used to treat various bacterial infections. The dosage depends on the infection type, severity, and patient condition…

Azithromycin is a macrolide antibiotic used to treat
Azithromycin is a macrolide antibiotic used to treat various bacterial infections. The dosage depends on the condition being treated, age, and medical history…
The dosage of Amoxicillin depends on the infection being treated
The dosage of Amoxicillin 250mg depends on the infection being treated, the patient’s age, weight, and medical condition. Below are general guidelines…
Capsules are a commonly prescribed antibiotic used to treat.
Amoxicillin 500mg capsules are a commonly prescribed antibiotic used to treat a variety of bacterial infections. It belongs to the penicillin group of….

